Introduction
Pharmaceutical companies are facing the increasingly-stressful challenge of delivering greater performance at less cost and in less time while complying with a rising number of regulations and guidelines (such as ICH GCP and the Clinical Trial Directive). This places tremendous pressure on the technical and managerial skill base of all companies, large, medium or small.
Training departments, clinical research specific trainers and managers responsible for developing clinical research staff are now faced with the need to deliver faster, effective, and lasting training solutions. These solutions not only need to eradicate existing gaps in skill, but they also need to build “World Class” competencies for clinical research staff.
Irrespective of the size of the company, resources may be limited for spending on training clinical research staff. Therefore, the investments in training need to be focused on providing valuable training solutions that strategically improve clinical research performance.
This limitation of resources may call out for the development and skilful execution of a pharmaceutical training strategy. This is not merely be a reactive solution by which personnel would enroll in publiclyavailable courses. Rather, it would entail building an internal training strategy that would be robust and resilient in the face of day-to-day pressures encountered while running clinical development programs. These pressures frequently overwhelm, and they sometimes wipe potential value from off-the-job training, as staff might not be able to fully implement what they have learned.
This article argues that a training strategy is imperative in order to retain and harvest the full value of key training interventions. Also, the training strategy needs to be appropriately sourced—not the easiest, most obvious solution on the market.
A “Training Strategy” can be defined as value-added, prioritized and readily-implementable plan for addressing both current and future performance gaps.
A training strategy links the organizational and business goals with the needs of individuals. The training strategy must be dynamic in order to account for changes within the organization as well as external changes (e.g., the increasing regulations affecting drug development such as the Clinical Trial Directive, and the ICH electronic common technical document, and increasing Phamacovigilance requirements).
The ingredients of this definition can be dissected as follows:
- Value-added – The training strategy should not only address the specific performance improvements that will be achieved, but it should also provide a business case for its rationale (e.g., the training intervention should be aligned with the clinical trial program development plan)
- Prioritized – The strategy should take into account relative attractiveness, difficulty of implementation, and urgency
- Implementable – It should be carefully tested against likely barriers to delivering its full value
- Plans – A coherent overview of key objectives, options and actions (the “how”), should be provided
- Thorough – The training strategy should not just be remedial – it should also address significant improvements in training skills, as well as gaps between current skills and new skills required to meet challenges in the future environment
Before we look at developing a training strategy, let us examine its potential added value.
“Value” originates from the idea of ‘economic value’; ultimately, all business activities have a point to directly or indirectly generate cash flow. Even value of business staff comes from having growth potential and security, and this is ultimately related to the cash flow.
Training from a variety of sources results in potentially-increased economic value.
In clinical research, this includes:
- Insightful and visionary decision-making in drug development (e.g., development of a lifestyle drug such as Viagra)
- Avoiding errors, disruption and re-work (e.g., effective monitoring for the reduction of data queries)
- Accelerating processes (e.g., faster patient recruitment through CRAs, more effectively motivating investigator site teams)
- Improving the adoption of technical know-how by the implementation of an electronic data capture system and a clinical trial management system
- Aligning behavior (e.g., clinical research staff and data management understanding each other’s needs to enable them to produce CFRs that facilitate more accurate data collection, as well as allowing easy data entry)
- Shifting mind-set (e.g., clinical research staff welcoming audit and inspection as an opportunity to verify the GCP compliance of their processes)
Each of these value-creating activities is targeted in terms of the ‘From-To’s’ of shifts – which will ultimately generate economic value. This can be expressed as a “From-To” or “FT” analysis (refer to Figure 1 for some examples of shifts). This analysis can be used to help assess the gap in expertise in a skills area in order to consider how to manage the gap and to help assess the effectiveness of the training intervention.
 Figure 1: From-To” or “FT analysis
Figure 1: From-To” or “FT analysisSome pharmaceutical companies are starting to assess the business value of implementing training strategies. This value-based mind-set is, perhaps, new to pharmaceutical companies and to those responsible for implementing clinical research training. Essentially, the crucial ingredient for every training initiative is to have a business case that approximately targets its value.
Developing a Training Strategy
Figure 2 diagrams a process for helping develop an effective training strategy. Here, we distinguish between:
 Figure 2: Stages of developing and implementing a training strategy
Figure 2: Stages of developing and implementing a training strategy- Diagnosis of current position
- Options
- Planning
- Implementation and roll-out
- Review of training
Diagnosis of current position typically begins with some focused competency (skills) analysis. Competency analysis involves collecting data and can easily become a major exercise, one in danger of becoming an end in itself. This is particularly true in the pharmaceutical industry, where there are many technical specialties.
A good starting point is to review the relevant current clinical research procedures to identify the primary clinical research job skills, task skills, scientific/technical and interpersonal skills. A range of approaches can be used to gather data, including structured interviews, questionnaires, focus discussion groups, audit findings, and formal or informal feedback including information from surveys about training needs. You do not need to use all these methods and often, a sample of structured interviews of both managers and staff regarding a particular job function provides very informative data, since this is a particularly flexible approach to obtaining insights. People are less likely to provide insights when simply answering a standard questionnaire.
The data collected will be reviewed and analyzed to allow you to evaluate the initial gap between the present competence and the skills required of a clinical research regimen. An initial training gap analysis may be drawn by distinguishing between the following:
- Where you are now, and where you need to be now
- Where you are now, and where you need to be in the future - to become what is considered “World Class” in clinical research
A report can then be produced to explain the purpose of the specific training needs analysis, the methods used to collect the data, the skills/ performance gaps and initial solutions for managing these needs. Some of these needs will be specific training solutions and others will be forms of organizational support, such as performance management.
Ideally, one would be “World Class” in everything regarding clinical research as well as in addressing all the gaps in skills identified from the training needs analysis. Not only is this unrealistic, it may also result in your spreading of training resources much too thinly.
As the training gap analysis is likely to identify a large number of training needs, it is very helpful to prioritize the areas to be addressed first in order to optimize effectiveness. It is useful to compare their potential attractiveness and implementation difficulty when carrying out an initial prioritization of training competency gaps. This can be achieved using a technique called the attractiveness-implementation difficulty grid (or ‘AID’ analysis) – refer to Figure 3 for some examples of possible training gaps. This output will vary depending on the particular findings of the training gap analysis, and will not be the same for all companies.
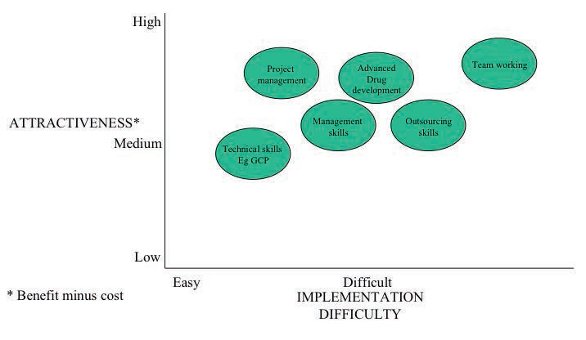 Figure 3: Attractiveness-implementation difficulty grid (or ‘AID’ analysis) for prioritising gaps in training skills
Figure 3: Attractiveness-implementation difficulty grid (or ‘AID’ analysis) for prioritising gaps in training skillsHere, it is invaluable to differentiate between continuous improvements (which entail maintenance of current capability, or moderate enhancements of it) and a major shift in capability that will have a major impact on current and future performance.
An example of a major pharmaceutical skills shift is that over the last few years, contract research organisations (CROs) have realized that they need to spend up to six months on intensive training to equip new untrained monitors with the skills to monitor clinical trials before they are capable of monitoring sites alone. This has enabled CROs to satisfy the pharmaceutical companies with the notion that they will achieve adequate quality standards for monitoring.
The area of electronic data capture (EDC) presents another example where some companies are looking to run 90% of their clinical trials using EDC in order to decrease the time to enter clinical trial data. This is a major change in clinical research and investigative staff work. For EDC implementation to be effective, major training initiatives are needed to facilitate changes in data collection processes.
A significant change that the industry is exploring is the development of management skills in clinical research international clinical trial programs. Additionally, the industry is examining clinical research for training the increasing number of clinical trial administrators, knowledge management changes in regulatory requirements, and clinical research accreditation.
Not many major training initiatives can be attempted simultaneously. Following the Japanese philosophy of breakthrough, or “HOSHIN”, it is best to implement only between one and three simultaneously, especially at the start of implementation. By staggering the effective implementation of major training initiatives over time, possibly five or six goals may be met in 18-24 months.
Once a number of strategic training priorities have been identified, it is essential to analyze their key interdependencies. All too often, training initiatives exist as relatively self-sufficient initiatives, while their full and potential value is highly dependent upon a number of actively-managed interdependencies. Achieving full alignment of these interdependencies requires considerable positioning and communication of each initiative with the key players involved. It is imperative to use a further visual method, such as thorough stakeholder analysis (refer to Figure 4). Here we see stakeholders (who might include decision makers, advisers, implementers, or recipients), analyzed by their respective attitudes (‘for’, ‘neutral’ or ‘against’) and their level of influence.
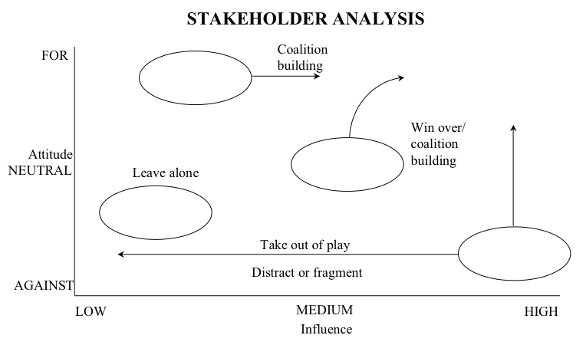 Figure 4: Stakeholder Analysis
Figure 4: Stakeholder AnalysisFrom my experience, all of the analytical techniques covered in this article must come into play in order to evolve an effective training strategy. The key training required in major areas of “stretch” and areas needing continuous improvement should be identified with the total involvement of senior line managers. Ideally, the training department should act as the specialist advisor on how training issues are best addressed, implemented and monitored. But this role should not wholly or primarily take over the training intervention.
Aside from obtaining the ownership of senior management from the very start, it is highly advisable that management is involved in the detailed planning, the implementation of the training programs, and learning the support processes themselves. The table below compares and contrasts the respective roles of line management and training department:
Reader exercise
- To what extent does the model of the various roles (as defined above) reflect practices within your organization?
- Where this does not happen, and what are the problems and costs arising as a result of this?
- Does line management currently have the skills and mind-set to accomplish this role? (If not, what training and support are required?)
- Does the Training Department currently have the skills, mind-set and credibility to achieve this? (If not, what training resources and repositioning are needed and should these be sought externally, either in whole or part?)
Unless there is a close and truly symbiotic relationship between senior management and the training department, then the default training strategy most likely to be implemented can be characterized as follows:
- A core of standard, regular in-house training programs which address past training needs – partially and as one-off courses without real follow-up and follow-through
- The value/ROI (return on investment) of training is frequently assessed primarily by “satisfaction sheets” rather than by genuine, tangible value-added assessment
- New needs are identified by senior management, and are frequently not well diagnosed. The Training Department/ training function is expected to react fast to put something in place, and this sometimes fails to deliver solutions
- Line managers and the training department/training function are reactive to individual requests for training, the result being that too much emphasis is placed on more expensive and untailored public programs. When individuals return to their companies, the learning dissipates as a result of the organization’s unchanged mindset.
Now, this brings us to the issue of sourcing your training strategy.
The above table illustrates that while standard public and incompany courses do have their place, a focus on them as being the primary source of training may be inappropriate and misplaced. Probably the best combination for training is that of tailored internal courses with follow-on project work and selective mentoring alongside a lower reliance on standard courses for predictable, core skills training.
A tailored, in-company approach is particularly suited for:
- Technical skills, whether these are company or department-specific
- Project management skills
- General management skills - including “people skills,” strategic thinking, change management, etc.
- Leadership and team-building
An example of a tailored in-course program which blends the best internal and external company perspective and addresses a major area where clinical research managers would benefit in improving their management skills is titled, “World Class Management Skills - The Example of the Mini Pharma MBA.”
Combining Internal and External Trainers
A very powerful delivery combination is to source tailored programs with an internal training source and an external provider who can add an outside perspective. The choice of an external provider can be based on the following criteria:
- Technical ability in clinical research and GCP, track record and credibility of training clinical research and GCP QA professionals
- Training skills and style
- Willingness to understand the company and deliver a trulytailored training solution
- Likely fit within the company’s culture
- Flexibility and responsiveness
- Focus on real value-added (rather than just on training and completing satisfaction surveys)
Conclusion and Next Steps
Pharmaceutical companies are putting ever-increasing demands on their staff to perform to “World Class” standards of clinical research, and this requires the development and implementation of an effective Training Strategy, utilizing the aforementioned sequence of identification, development, and implementation. Developing a truly robust strategy requires the symbiotic work of senior managers and a business-focused, strategic training department.
The training strategy should contain a number of key (prioritized) major training initiatives at its heart. These should be diagnosed with line managers, be well positioned, and should then be sourced in an innovative way to fulfill needs. Training solutions should be as tailored as possible, preferably with less emphasis on public courses and standardized internal courses.
Developing a training strategy is an exciting frontier for bringing clinical research into line with world class management. Is your company ready for the challenge?
Dr. Laura Brown, MBA is an independent training consultant and is Course Director of the M.Sc., School of Pharmacy at Cardiff University in England. She was formerly head of a training company delivering international public courses. She now specializes in developing training strategies, competency assessments, and the delivery of project management, clinical research courses and management skills programs (including the in-company mini Pharma MBA).