Introduction
Since the early 1980s, biotechnology products have shaped the pharmaceutical industry. A large number of monoclonal antibodies and therapeutic proteins have been approved and are anticipated to be a major growth driver for the industry in upcoming years [1-6]. Mammalian cells are the expression systems of choice due to their ability to properly fold and modify these complex proteins therapeutics [7-10]. Tremendous advances in process development throughout the last decade have resulted in significantly increased manufacturing scales, product titers, and recovery yields thereby satisfying increasing market demand for existing and new products. Manufacturing scales up to 25 m3 operated in batch, repeated batch, or fed batch mode followed by a sequence of chromatography, filtration, and concentration steps represent state-of-theart technology [11, 12] – a typical antibody production process is shown in Shukla and Thömmes 2010 [13].
However, the industry will face numerous challenges in the coming years. A review of data from the 1950s to 2008 indicates a trend toward stagnant R&D outputs and significantly increased development cost, representing a major threat for most innovation-driven pharmaceutical companies [5]. In response to this threat, several measures to improve competitive capabilities along the value and supply chains have been successfully implemented (e.g. lean and six sigma principles). Biopharmaceutical manufacturing in the 21st century must be agile, nimble, and flexible in order to adapt quickly to a changing environment and guarantee uninterrupted supply of high quality therapeutics with acceptable financial performance. Continuous improvement in manufacturing efficiency will enable biopharmaceutical organizations to divert resources toward R&D investment, thereby making manufacturing externally supportive to the overall corporate innovation strategy [14]. In order to satisfy the demand of existing and new products, investments in process development have resulted in a large variety of new technologies that have made their way into manufacturing. Most companies have established platform technologies to accelerate timelines and avoid excessive investments into existing manufacturing assets. Optimal manufacturing costs, asset utilization, and supply chain risk mitigation are key success factors in delivering value to the industry. Therefore, the management of technology plays a key role in the continuum of R&D to full scale manufacturing (e.g. incremental innovation, technology transfers, etc.) [15]. In a global manufacturing network with diverse assets and needs, it is both critical and challenging to address today’s requirements while preparing for tomorrow’s needs. If not managed properly, the benefit and efficiency of (incremental) innovations driven out of R&D might not be utilized to their maximum potential resulting in lost opportunities and waste in commercial manufacturing. In order to increase efficiency, operational excellence (OE) programs in existing business processes (BP) and manufacturing processes have been developed (lean for waste elimination, Six Sigma for variability reduction).
These techniques have enabled manufacturing across different industries to deliver the existing product portfolio at lower costs and higher quality. The pharmaceutical industry is still in the early stages of becoming like Toyota in the automotive industry, but it is moving in this direction [16- 19]. A more strategic approach is required to optimize resource utilization and minimize investment [14, 20]. Taking advantage of new technologies, such as disposables, new purification resins, and different operation modes that increase production yields, streamline processing steps, and lower costs, can provide companies with a critical competitive edge.
Several methods and tools have been discussed to support proactive technology management. For example, integration of manufacturing requirements at an earlier stage in the process of product development enables a smooth transfer of new products and technologies. On the one hand, this approach is more resource and staff intensive. On the other hand, it reduces both investments and lead time in later stages of technology implementation. Integrated product and process development (IPPD) has been described in the literature for several years and is just one approach to avoiding cost and lead time increase [21-23].
Another way of increasing performance is continuous improvement of existing technologies and manufacturing processes. During the product life cycle, there is an opportunity to apply mature technologies driven out of R&D, to existing products, assets, and processes resulting in quality improvements, supply chain risk mitigation, and increased robustness.
This article describes the foundation and success of a global Biologics Drug Substance Manufacturing Technology Council operating at the interface of strategic planning, supply chain and process development whose charter is to manage current and future production challenges on a global network and a manufacturing site level. The work describes organizational elements and tools utilized to derive a networkwide technology roadmap including a “technology innovation architecture” (an adaptation of the Innovation Architecture published by Sauber 2003 [24]). This roadmap encompasses the assembly of a prioritized network portfolio leading to efficiencies, cost savings and process improvements. This work provides answers to the following questions:
- How should manufacturing anticipate future demands, required technologies and capacities?
- What is a suitable way to analyze current assets, technologies and manufacturing competencies in terms of fit for existing products and future products?
- How can business-critical innovations driven out of development and incremental innovation driven out of manufacturing be aligned to optimize existing assets and technologies to drive down costs and lead times?
- How can existing teams and processes be aligned and utilized to enable enhanced management towards future needs and demands?
Analyzing the Status Quo
The global Biologics Drug Substance Manufacturing Network, Hoffman-La Roche AG, Switzerland, evolved by several investments in the construction of new facilities, as well as mergers and acquisitions. To manage this network, several positive elements were employed by the company. Best practice teams for several process unit operations were established in the network. A substantial knowledge base from R&D to commercial manufacturing existed, and several technologies were applied in different ways in the organization. However, in most cases clear priorities, reporting lines, and global alignment were missing. Clear coordination was lacking, leading to technology implementation redundancies, and non-optimal usage of network resources. In order to deliver user requirements to the process development organization aimed at improving manufacturing processes, as well as to create a feedback loop after implementation of new technologies, a group of network-based subject matter experts from Technology, Manufacturing and Process Development was formed to be integrated into the process development process. A strategic innovation management team was deployed to drive the project portfolio for incremental innovation in manufacturing. As a result, two different global teams and separate business processes were established to manage a portfolio of projects leading to redundancy and misaligned utilization of resources across the network. Although all projects, initiatives, and activities targeted individual issues and opportunities, a holistic strategic roadmap towards short term and long term needs did not exist. In addition, the organizational decision making processes often did not find the right audience to drive management attention and establish distinct network priorities and goals.
Several other business processes, including site selection for new products, knowledge management, agile resolution of manufacturing issues, global inspection response management, etc., existed but showed the need for a better management model to enhance efficiency and foster alignment in the global manufacturing network. These issues manifested themselves as “symptoms” that included sub-optimal resource utilization, nontargeted investments, and missing competencies in the existing workforce. These symptoms led to reactive behaviors rather than the proactive management of technology, assets and workforce. Clearly, these issues needed to be resolved to develop a more strategic approach toward integrated management of technologies and innovation.
Four Steps to Excellence in Technology and Innovation Management
Step 1: Implementation of an Aligned and Streamlined Team Structure in the Organization
The literature discusses several models for technology and innovation management. In general, these models are designed to serve at a high level and focus on the exploration of new business fields. Rohrbeck and Gemuenden 2006 [25] describe some organizational elements and functions of innovation management for different companies. Novartis described a system called “future watch”, which consisted of technology scouts looking into future trends and business opportunities. Deutsche Telekom AG implemented a so-called “technology radar” to observe technology developments globally. Volkswagen AG described a project called “Moonraker” that brought experts to a house in Malibu to analyze and work out strategies. Siemens designed an integrated concept called “picture of the future” to draw potential future scenarios in cross functional teams. Some companies have implemented a Chief Technology Officer (CTO) at the board level to manage technology and innovation, increasing management awareness and providing a single function responsible for technical competitiveness. However, these methods and organizational constructs are primarily used to open new markets, develop business fields, or drive early product development in research. For manufacturing, this is not necessarily the right approach since it is resource intensive and fails to address the operational focus of production. There is, however, an opportunity to use some of these elements to link upcoming product portfolio developments and industry trends to existing products, technologies, assets, and production technologies in order to create organizational awareness of current and future needs.
An interesting model was published by Tschirky, 2003 [26]. Here several permanent and non-permanent elements were combined in a technology management strategy for the Human Tissues Corporation. Of specific interest was a central technology group reporting into a person accountable for technology management (CTO), cross functional teams of experts and non-experts, communities of practice, knowledge management, and business intelligence.
Some of these elements already existed in Roche’s global Biologics Drug Substance Manufacturing Network, but needed to be redesigned. In order to analyze the current status and to achieve maximum efficiency, all existing teams and initiatives, including their goals, team charters, organizational setup, project portfolio and goals, were analyzed. In order to minimize resistance, a “founder” team was created with representatives of all key stakeholders and leaders of the several existing teams. This approach gained insights into the mission and vision of each team, as well as their degree of resistance to change. The founder team reported into the ultimate decision-maker – a member of the drug substance manufacturing leadership team accountable for technology management. The single decision maker at the highest level of the organization was important to both avoid bypassing decisions and to capture the attention of senior management and focus the organization. After the inventory of current efforts was completed, a team charter, including tasks, missions and goals, was established. This data-driven approach facilitated the identification of obvious overlaps and fostered discussions with senior management on the future role, tasks and mission of the new organizational function. Constant feedback and check-in at crucial points of the process with senior management to clarify direction, derive necessary decisions, and restate direction, was helpful in maintaining momentum for all involved parties.
As a result of this effort a new organizational element - the global “Biologics Drug Substance Manufacturing Technology Council” – was created (see Figure 1). This organization included three sub teams: Manufacturing Process Evaluation Team, Engineering Network Team and Validation Network Team.
 Figure 1. Schematic Structure of the Biologics Dugs Substance Manufacturing Technology Council (SME = Subject Matter Expert; MSAT=Manufacturing Science and Technology)
Figure 1. Schematic Structure of the Biologics Dugs Substance Manufacturing Technology Council (SME = Subject Matter Expert; MSAT=Manufacturing Science and Technology)In order to drive effective decision making, the senior management team of the drug substance manufacturing network consisting of the manufacturing site heads, strategic planning, supply chain management, process development, quality function, finance and human resources, served as the steering committee for the Technology Council core team. The steering committee endorsed the projects, initiatives and budgets for the council which were subsequently integrated into the manufacturing site budgets across the global network.
The core team consists of site functions accountable for technology and manufacturing science, engineering, quality, strategic planning, and process development in order to drive a holistic perspective and decision making. The clear advantage of integrating the local manufacturing sites is to foster a bottom-up and top-down approach. The primary tasks of the core team are as follows:
- Provide a unified perspective on operational strategies
- Manage future needs by implementing a clear technology roadmap
- Develop an associated portfolio of projects and initiatives
- Provide a framework for sharing best practices
- Act as a single point of contact for emerging issues to other functions
- Achieve global alignment on business and manufacturing processes
- Align inspection responses, as well as corrective and preventive actions
The sub-teams mentioned earlier reported into the core team for guidance, decision making, and endorsement of team activities or proposed task forces for addressing emergent issues.
In order to clarify intent and create transparency at the interface with R&D, the team aligned roles and responsibilities in the technology management process. A simple technology lifecycle management model was established and agreed to. After completed feasibility assessment of new/mature technologies in R&D, the first implementation into a commercial manufacturing facility was performed under the governance of the council. Further application for continuous improvement was guided and assessed by the council. This helped free up resources in R&D and gave clear direction to the manufacturing and technology organization in the commercial manufacturing facilities. Because all improvements and projects were assessed through the global teams, an alignment of changes, assessment of needs and risks, as well as streamlined portfolio management was guaranteed. For example, regulatory and quality implications were assessed similarly and all manufacturing processes and enabling processes could be kept aligned – a prerequiste for multisourcing in a global manufacturing network.
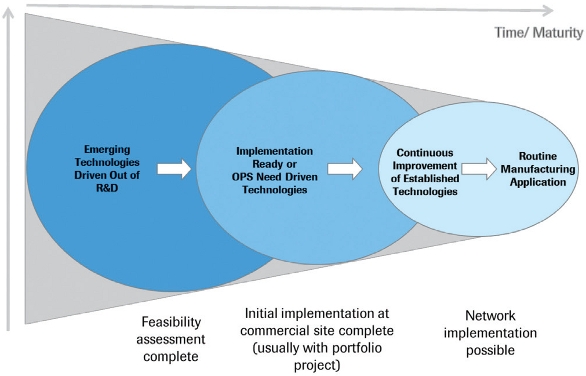 Figure 2. Simple Technology Lifecycle Management Model
Figure 2. Simple Technology Lifecycle Management ModelThe implementation of this structure led to some quick wins and efficiencies, which can be described as follows:
- Simplified team landscape
- Minimized resource requirements by consolidating teams and avoiding redundancies
- Review of ongoing project portfolio and stopping non-targeted activities
- Aligned goals, mission and deliverables of the teams
- Facilitated agile, nimble and efficient decision making and feedback to initiatives in other parts of the company
- Aligned network goals and site goals on a global level
- Created a clear governance structure and decision making
- Defined touch points, responsibilities and scope for internal and external customers
- Created ownership of a targeted roadmap to provide guidance for the network on projects, initiatives and needs
Step 2: Creating a Technology Roadmap
In order to develop an end-to-end view of future needs, a business process was designed to drive desired outcomes on an operational strategic perspective of 0-5 years along with an outlook for years 5-10 (see Figure 3). A vital part of the strategy setting exercise was the design of a global technology roadmap.
 Figure 3. Business Process Set to Execute Strategy
Figure 3. Business Process Set to Execute StrategyTechnology road mapping is a widely-used technique in several industries for aligning long-term strategies with markets, products and technologies [26-28]. The roadmap allows for the identification of priorities and opportunities for investment (including markets, products, technologies, processes and people). In addition, it is a structured method to align future goals/needs and critical activities in the organization. Several definitions, applications and levels of detail have been published. Road mapping can generally be described as a process or tool to analyze today’s realities and describe a path forward (or strategy) towards future requirements in an integrated way [26-30]. The output of the process is a document that shows aligned priorities, needs and actions – depending on the granularity of the roadmap. For a manufacturing network it is important to analyze the environment – however a market analysis for business fields is not necessarily of high importance. In addition, readiness for future product portfolios driven out of R&D, as well as the application of emerging technologies to improve efficiencies, safety and compliance in current assets/processes is of vital importance.
The success of roadmaps depends on several factors:
- relevance of topics and business need;
- involvement of the right people and sponsorship on an executive level;
- clear process to set up the roadmap;
- and sustainable follow-through in execution to deliver (Albright und Kappel 2003 [30]).
As a first step in setting the direction of the group, an input/output analysis of processes was designed and a stakeholder analysis was performed. A detailed analysis of suppliers of, inputs into, outputs from, and customers of the roadmap activity was crucial to enable efficient and targeted delivery. In addition, the identification and involvement of key stakeholders/customers was crucial for later implementation and acceptance in the organization. One major requirement to set up a strategic technology management process is detailed knowledge regarding internal products, technologies, product pipeline, capabilities and competencies, trends and upcoming innovations in the business environment. In this case a portfolio analysis, expert interviews, lead user panels, and benchmarking were chosen to identify a path toward future requirements and demand based on current technologies, functions, competencies, and capabilities.
A voice of the customer analysis was performed that included site representatives, enabling functions and senior management to identify expectations, future needs, links to other processes, trends as well as strength and weaknesses. In addition, current site capabilities, technologies, and workforce competencies, along with current and future product characteristics and required technologies were analyzed in a product/site capability matrix. Benchmarking was performed by external analysis, e.g. conferences, and external expert interviews.
Based on this input, the next step in developing the technology roadmap was to design a strengths, weaknesses, opportunities and threats (SWOT) analysis to identify focus areas and gaps for future needs and demands. A modified version of the “Innovation Architecture” published by Sauber 2003 [24] was utilized to identify current capabilities and future needs. Instead of looking into innovations, fields, markets and business to identify future markets or products, a “Technology Architecture” was designed that assessed trends in the manufacturing environment to be ready for the future (such as product portfolio, product titers, regulatory expectations, quality requirements and trends, process platforms, supply chain long range plans, data analysis, trends and tools, etc.). Here the close relationship and integration between process development, manufacturing, quality and strategic planning as outlined in the governance structure were key. Since details are confidential only a schematic figure is shown in Figure 4. The details and granularity of the Technology Architecture varies depending on task and needs.
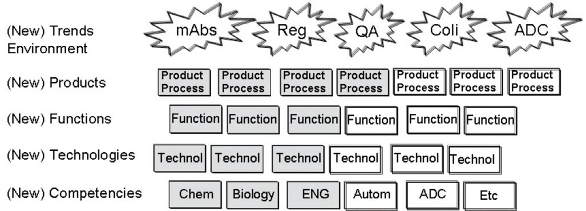 Figure 2. Simple Technology Lifecycle Management Model
Figure 2. Simple Technology Lifecycle Management ModelThis approach enabled a structured, comprehensive, and streamlined discussion around future needs. Surprisingly, the majority of these needs and the resulting gaps were driven from environmental trends and not by the product portfolio itself. Hence, a holistic view and the input of support functions and other processes (especially long range processes for supply planning, technical development portfolios and regulatory trends) are of crucial importance to drive decisions and future activities based on an end-to-end view.
As a final step, detailed activities and project charters for the identified gaps, focus areas, and initiatives were developed, including resource requirements and timelines. Clear prioritization resulted by using previously aligned goals in connection with overall network goals. Figures 5 and 6 describe the process in a simplified manner.
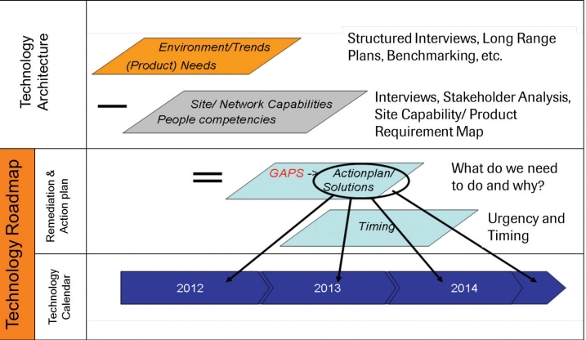 Figure 5. High Level Process to get from the Technology Architecture to a Technology Roadmap/ Calendar.
Figure 5. High Level Process to get from the Technology Architecture to a Technology Roadmap/ Calendar.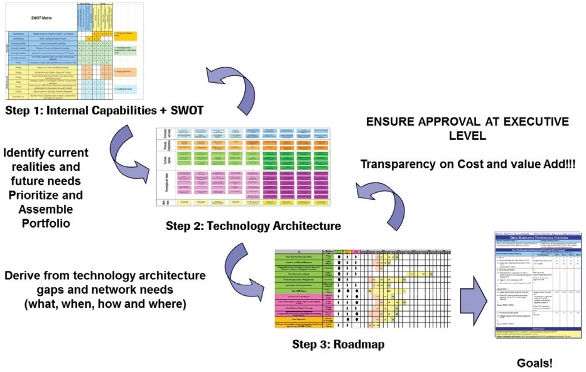 Figure 6. Overall Process from SWOT to Local and Global Goals
Figure 6. Overall Process from SWOT to Local and Global GoalsStep 3: Link to Other Processes and Roll Out
The technology roadmap is an active document so the link to other business processes is required. In addition, the implementation and approval of budgets and resources, as well as the impact to overall network capabilities and competencies, is crucial to avoid disconnects and untargeted development. Since the environment – especially demand figures and success rates of a portfolio pipeline – can change, a link to long range supply planning is necessary. It is crucial to drive local and global connections as the impact on budget is primarily on a manufacturing site level, for example via long range capital plans and site business plans, to accommodate the roles and responsibilities of the site project portfolios. In this case, four major processes were linked (see Figure 7).
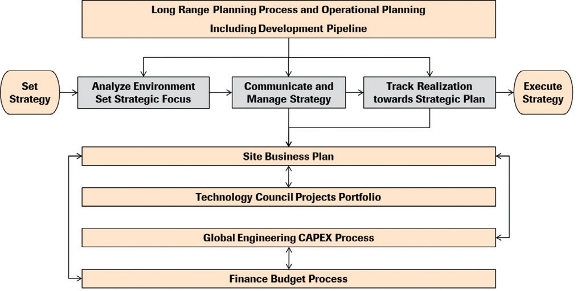 Figure 7. Link to Other Business Processes.
Figure 7. Link to Other Business Processes.The supply long range plan, including the development pipeline, was an input into the roadmap design. By linking the endorsed roadmap and project portfolio to the site business plans (owned by the manufacturing sites), all activities and investments are automatically linked to the budget and capital expenditure process allowing smooth execution, planned and predictable performance and clear prioritization on a global and site level for this subset of projects and initiatives.
Once established, it was crucial to roll out the concept and derive support within the organization and the support functions. Next to roll out and go live presentations, web- based tools facilitating decision making transparency, providing progress updates and illustrations of quick wins were necessary for depicting performance and managing change in the organization. Maintaining, updating, and adapting processes, infrastructure, and the roadmap to a changing environment was crucial to gain trust and momentum in the organization.
Achievements and Lessons Learned
Several achievements were made from a short, mid and long term perspective. Along with the streamlined, simplified and more structured organizational set up, the targeted utilization of resources towards future needs has driven significant improvements.
The Technology Council serves as a single point of contact for fast and agile implementation of corrective actions to improve compliance and robustness of the manufacturing network. In addition, shared and aligned best practices to increase standards in the company have added value to compliance, robustness and efficiencies.
A more streamlined site selection process was implemented allowing agility and transparency in decision making as a network with a more holistic end-to-end perspective on capacity utilization, network capabilities and portfolio mix at a network and manufacturing site level. Network-wide troubleshooting activities were controlled and guided through the council and competency centers/communities of practice leading to accelerated improvement of several manufacturing processes – one improvement, for example, led to a productivity increase of approximately 10% in one of the major facilities benefiting the company, the network and, of course, the patients.
From a mid and long term perspective, the assembly of a manufacturing technology roadmap helped to significantly streamline the project portfolio and focus the resources to network priorities. Driven out of a structured analysis of the environment, portfolio, supply demands and capabilities, a technology architecture was designed illustrating major needs and the execution of necessary projects and solutions.
As an example, the design of a common automation platform is being performed at one site and, as mutually agreed upon by Council, the solution will be transferred to the other manufacturing sites in the global network. The NPV on this project was calculated to be $17 million. The identification of redundant activities at the site level acted as another example. These activities have been stopped enabling several full time equivalents to work in a more productive manner.
The improvement of product changeover (PCO) times was identified as a potential cause of capacity issues during the analysis. With the growing product portfolio, a potential downtime due to PCO in the global network was assessed to be approximately 200 days. By sharing best practices and having a clear project plan these times could be minimized by 80% resulting in a gain of 160 days of production capacity.
Several other examples of introducing mature technologies for supply chain risk mitigation including streamlined data analysis technologies to improve process controls and disposable technologies were delivered to create meaningful cost savings. However, the biggest gain was the targeted cross functional-cross site approach that created efficiencies and transparency to manage proactive future needs as well as aligning processes across the global manufacturing network.
Lessons Learned
The majority of the implementation was effective, however, cultural change needs to be managed carefully. A global, rather than local, mindset requires different behaviors and thinking. A portfolio itself can be effective in theory, but stopping activities locally for the good of the global network can become challenging in practice. In addition, the discipline to execute the roadmap and to prioritize aligned requirements is of crucial importance. A major enabler for the overall structure and decision making is the support and clear commitment of senior management. This is also valid for each of the activities and projects. Sponsorship from the core team and/or relevant stakeholders helps to avoid pitfalls on the way to success. The detailed analysis and definition of desired inputs and outcomes was slow at the beginning, but enabled higher velocity at the end due to clear goals, processes and structure. Overall, the approach to deploy tools that kept the roadmap simple and transparent made the difference.
Acknowledgements
The authors acknowledge the members of the Biologics Drug Substance Technology Council, especially Harry Lam. Rod Franklin and Marco Jenzsch are acknowledged for editorial support.
References
- Reichert JM. Monoclonal antibodies in the clinic. Nat Biotechnol. 2001; 19:819–822.
- Reichert JM. Therapeutic monoclonal antibodies: Trends in development and approval in the US. Curr Opin Mol Ther. 2002; 4:110–118.
- Reichert JM, Pavolu A. Monoclonal antibodies market. Nat Rev Drug Discov. 2004; 3:383–384.
- Drapeau M, Sullivan F, Moniz Carpenter J. Special Report: Blockbuster Then and Now-Trends for Billion-Dollar Drugs. Spectrum Therapy Markets and Emerging Technologies. 2007; 12:1–39.
- Munos B. Lessons from 60 years of pharmaceutical innovation. Nature Rev. 2009; 8:959–968.
- Pohlscheidt M. Strategic Technology- Network-and Innovation Management. MBA Thesis. 2010; ETH Zurich, Switzerland.
- Xie L, Wang DIC. Integrated approaches to the design of media and feeding strategies for fed batch cultures of animal cells. Trends Biotechnol. 1997; 15(3):109–13.
- Qi HN, Goudar CT, Michaels JD, Henzler HJ, Jovanovic GN, Konstantinov KB. Experimental and theoretical analysis of tubular membrane. Biotechnol Prog. 2003; 19(4):1183–1189.
- Wurm FM. Production of recombinant protein therapeutics in cultivated mammalian cells. Nat Biotechnol. 2004; 22:1393–1398.
- Sethuramann N, Stadheim TA. Challenges in therapeutic glycoprotein production. Curr Opin Biotechnol. 2006; 17(4):341–346.
- Heath C, Kiss R. Cell Culture Process Development: Advances in Process Engineering. Biotechnol Prog. 2007; 23:46–51.
- Su WW. Bioreactors, Perfusion. Encyclopedia of Cell technology. 2003; 978 – 993. John Wiley & Sons. Inc.
- Shukla A, Thömmes J. Recent Advances in Large-Scale Production of Monoclonal Antibodies and Related Proteins. Trends in Biotechnol. 2010; 28:253–261.
- Wheelwright, SC, Hayes, RH. Competing Through Manufacturing. Harvard Business Review. 1985 January/ February: 2-12.
- Pohlscheidt M et al. Pharmaceutical Outsourcing. Avoiding Pitfalls During Technology transfer of Cell Culture Manufacturing Processes in the Pharmaceutical Industry – Mitigating Risk and Optimizing Performance 2013 14(2):34-48
- Correa HL. Agile Manufacturing as the 21st Century for Improving Manufacturing Competitiveness. In Editor Gunasekaran A Agile Manufacturing as the 21st Century for Competitive Strategy. Kidlington, Oxford, UK: Elsevier Science Ltd 2001:3-24
- D´souza A, Keeling, DJ, Phillips, RD. Improving Quality in Pharma Manufacturing. The McKinsey Quarterly - Health Care Sep 2007
- Cremer P, Loesch, M, Schrader U. Maximizing Efficiency in Pharma Operations. The McKinsey Quarterly - Operations Practice 2009: 2-4
- Bhadoria, V, Dedeurwaerder T, Tilley J. Holding the diagnostic mirror. Mc Kinsey Quaterly 2010. pp 1-6
- Hayes RH, Wheelwright, SC, Clark, K. Dynamic Manufacturing Creating the Learning Organization. Free Press, New York, NY, USA, 1988
- Schrage DP, Mavris, DN. Integrated Product-Process Development (IPPD) Through Robust Design Simulation (RDS). In Editor Gunasekaran A Agile Manufacturing as the 21st Century for Competitive Strategy. Kidlington, Oxford, UK: Elsevier Science Ltd 2001:95-112
- Pisano G. Eli Lilly and Company: Manufacturing Process Technology Strategy, Teaching Note 9-692-056. Harvard Business School, Harvard University, Boston, MA, USA . October 1998.
- Burt, JA. Department of Defense USA; Guide to Integrated Product and Process Development, Version 1.0, Office of the Undersecretary of defense, Washington DC, USA, 20301, 1996
- Sauber, T. The Innovations Strategy Development Process Based on Innovation Architectures. In Tschirky H, Jung HH, Savioz P. Technology and Innovation Management on the Move – From Managing Technology to Managing Innovation driven Enterprises. Zurich Orell Fuessli. 2003
- Rohrbeck R, Gemuenden HG. Strategische Fruehaufklaerung – Modell zur Integration von markt- und technologieseitiger Fruehaufklaerung. Vorausschau und Technologieplanung 2006 (Presentation http://www.rene-rohrbeck.de\)
- Tschirky H. The concept of Integrated Technology and Innovation Management. In Tschirky H, Jung HH, Savioz P. Technology and Innovation Management on the Move – From Managing Technology to Managing Innovation driven Enterprises. Zurich Orell Fuessli. 2003
- Kappel TA. Perspectives on roadmaps: how oganizations talk about the future. The Journal of Product Innovation Management. 200: 18:39-50
- Rinne M. technology Roadmaps: Infrastructure for Innovation. Technology Forecasting and Social Change. 2004;71:67-80
- Phaal R, Farrukh CJP, Probert DR. Technology Roadmapping – A planning framework for evolution and revolution. Technology Forecasting and Social Change. 2004; 7:81-100
- Albright RE, Kappel TA. Roadmapping in the corporation. Research Technology Management. 2003; 46:31-40
Michael Pohlscheidt received his degree in bioengineering in 2001 from the University of Applied Sciences in Aachen, Germany. His Ph.D. thesis was performed at Bayer HealthCare AG and Bayer Technology Services and guided by the University of Magdeburg, Germany (2005). From 2005 to 2010, he worked in different positions at Pharma Biotech Production & Development, Roche Diagnostics GmbH, Penzberg, Germany. Since 2010, he has worked at Genentech Inc., Oceanside, CA, first as an associate Director Manufacturing Science and Technology and since 2012 as Director of Manufacturing Operations.