Introduction
The research-based pharmaceutical industry has always accommodated change, and like other businesses, is increasingly becoming cost constrained due in part to the rising investment required to develop new medicines and the global economic environment. Although greater numbers of new drug candidates are being identified for development, personnel as well as capital resources have become increasingly constrained to conduct all aspects of development in-house. In order to make the best use of available skills, many of the traditional functions are now being outsourced or selectively integrated, to enable existing in-house resources to focus on activities which provide proprietary knowledge. In recent years, cost efficiencies have been made, typically through outsourcing to contract research organizations (CRO’s) located within close geographical proximity to the industrial sponsor. Advantages to a CRO being local to the sponsor included the ease of communication within a shared timeframe and travel between sites being relatively simple. However, with the advent of emerging technological capabilities in countries where not only are there an abundance of qualified scientists, but the infrastructure costs and compensation are relatively low as well, the incentive to conduct work in “low cost countries” (LCC), outweighs the advantage of being in a nearby location. Although the same principles apply to outsourcing, irrespective of the geographical location, this paper addresses some of the challenges and experiences which Bristol-Myers Squibb (BMS) have overcome to outsource Long Term Stability Studies (LTSS) to low-cost country CRO’s, and highlights some pitfalls which should be avoided.
Decision Process to Determine Project Suitability for Outsourcing
The first decision to be made in placing an LTSS at a low-cost country CRO, will be based on acceptable technical skill set, capability/infrastructure, quality and compliance track record, safety and timeframe criteria. An example decision tree is shown in Figure 1 and can be used to determine the suitability of placing a project overseas or retaining the study in-house.
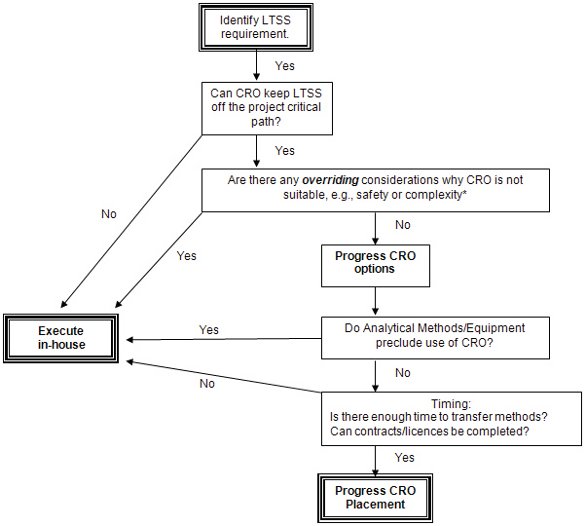
Figure 1 - Typical decision tree to determine suitability for LCC outsourcing of LTSS.
Specific Challenges and Considerations to LCC Outsourcing
Cultural Differences
It is an advantage to identify a LCC contract laboratory that is fluent in the native language of the sponsor company and has trained staff and business processes in place, which is friendly to Western operating style. Alternatively, the sponsor laboratory could have a primary contact who is fluent in the local spoken language of the selected vendor. This enables clear communication of expectations and facilitates flawless method transfer and unambiguous troubleshooting when unanticipated events arise. Additionally, familiarities with the geography, local business practices, regulation and management style are important in ensuring smooth and sustainable endeavour.
Transport, Customs, Import Licences, Test Licences
For a typical study, large numbers of samples will require transporting to the LCC contract laboratory. These can be provided in bulk containers for packaging at the contract laboratory or alternate local facility, or can be provided in final packaging for immediate storage under the test conditions. In either situation, it is likely that samples will require shipping under controlled and monitored conditions to ensure the products are not exposed to detrimental environmental conditions such as high temperature and humidity for long periods of time. The use of environmentally controlled containers which have integral temperature monitors is critical as conditions external to the shipping container may become extreme, e.g., airport warehouses, airplane storage holds, customs holding areas, local road transport delivery vehicles. Careful planning is essential with couriers to avoid delays in the export/import process due to Customs border control processes. It may also be necessary to obtain Import licences and testing licences from national or local government authorities. The contract laboratory should be able to provide advice on these requirements, and authorization should be requested in adequate time to initiate the study to meet business requirements. BMS’s experience is that the time to ship samples from the sponsor site to the CRO laboratory takes an average of 22 days (range 14-37 days) and this should be factored into timelines.
Audits
The auditing process is a key tool in ensuring compliance with both GMP and sponsor practices. Being geographically remote, LCC’s will generally incur higher travel and time costs when compared to local contract laboratory audits, so careful planning on the content (with emphasis to address potential vulnerabilities) and timing (to coincide with key stability time points) is critical.
Project Selection
Forming a strategic alliance with a contract laboratory in a LCC will require the development of trust and confidence from both partners. Working with a remote CRO in a different time zone will require a higher level of scrutiny until confidence has been established. To build that confidence, it is recommended that an initial mock study or an actual “low-risk” initial study be conducted. Such projects would include those not on the critical path to a regulatory submission, those with low-medium technical challenges, possibly a back-up to a primary candidate, and which has no intellectual property risk.
Such a “low-risk” study will help determine the capability of the CRO to a) perform testing which includes sourcing analytical method consumables, b) data capture, review and storage, c) overall quality systems assessment and d) stability chamber and sample management.
The method transfer process which follows should include an assessment of the availability of method consumables such as grades of reagents, filters, chromatographic columns, etc. Cost saving opportunities may arise to source local equivalent consumables which have been appropriately qualified for use. This would obviate the need to supply consumables from the sponsor lab, incurring additional transport costs.
Reporting of Data (Including Raw Data, e.g., Chromatograms)
Agreements should be made on the timing and format for the provision of approved test data and the level of scrutiny from the CRO QA function. It is likely that the sponsor laboratory will have SOP’s which define the maximum period to complete testing and report values, and these should be included in the service agreement. If data is to be entered into the sponsors LIMS system (or equivalent) informatics solutions will be required which are compatible with systems of the sponsor, e.g., capability of data networks, which may be provided by a service provider in the LCC and may not have equivalent performance to the country of the sponsor.
Agreement should also be reached on the retention location of primary data, e.g., chromatograms, lab notebooks, which may be required for review at the time of a regulatory inspection. During the CMC submission review process, the reviewer may request to see actual data supporting the stability information filed in the dossier. This may not be a challenge if the reviewer is prepared to travel to the LCC, or if the data is accessible electronically, e.g., using a web-based system. However, consideration may be required to ship data or exact electronic copies thereof, back to the sponsor laboratories prior to a regulatory inspection (e.g., PAI). Agreement should also be reached upfront to cover requirement to have hard copies of representative chromatograms at selected stability storage conditions, time points for packages and product strengths available for submission to certain Health Authorities as part of the commercial approval process.
Fee for Service versus Strategic Alliance
There are different strategies which can be employed when considering outsourcing to a LCC. One approach is to treat each study as being independent (fee for service) in which case, the sponsor will be required to conduct a rigorous selection and audit process to generate confidence in the data being generated for every study. Another approach is the development of a strategic alliance between the sponsor and a single contract laboratory. This will have added benefits as a result of familiarity with personnel, practices and procedures. A reduced number of quality system audits will need to be conducted and cost advantages should be offered as multiple project assignments confirm business continuity. Should the sponsor and contract laboratory develop such a partnership, the same analysts may be employed in a flexible manner to ensure continuity of experience associated with working practices of the sponsor methods. A variant of this approach is to employ contract staff at the CRO who are dedicated to work only on the sponsor’s projects and who would be available to perform other analyses (not specifically LTSS, e.g., process justification sample testing) as directed by the contract provider.
Steps to Successful LCC Outsourcing
Developing a Process Model
The outsourcing of stability studies, particularly to an offshore CRO in a LCC, will only be successfully achieved if the process is considered a partnership between all contributors and not just a financial transaction. A partnership where the CRO laboratory becomes an “extention” of the sponsor’s own lab operation is a most desirable approach versus CRO scientists working as “pairs of hands.” Clear delineation of roles and responsibilities early in the off-shoring process is critical to ensure success.
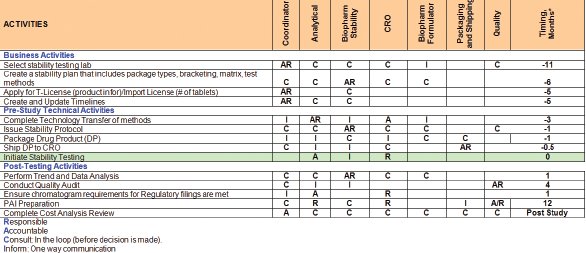
Figure 2 - Example RACI Chart for LTSS outsourcing to LCC.
The sponsor will initially have subject matter experts (SME) within different functional areas. It is imperative that at an early stage of the project, clear expectations and accountabilities are assigned to all SMEs who should have a working understanding of the entire process whilst retaining expert knowledge for their specific role. Communication between the sponsor and vendor will be expedited if both parties identify a coordinator for the study, and these individuals can liaise regularly with each other and act as a point of contact within their own organization. Their role will be as project managers for the study. Within BMS, we have assembled a series of RACI charts to enable each project manager/ coordinator and SME to identify who has Responsibility/Accountability/Consult/Inform role within the process. Each task within the LTSS outsourced sequence of events has been assigned a RACI designation for each functional area. This also acts as a checklist and process flow for the many activities required for a successfully outsourced LTSS. An example of such a RACI chart is given in Figure 2. The example given illustrates a high-level process flow, which can be further categorized to give a greater degree of detail. As the LCC contract lab is likely to be considerably remote to the sponsor site, it is important to review the process model with the vendor and ensure they fully understand their responsibility and accountabilities and that there have been no misunderstandings arising from language barriers.
Selecting a Country and CRO
There are several primary considerations when selecting any CRO. The primary requirement which is non-negotiable is the Quality/Integrity of the data. Other critical considerations include the technical ability and capacity of the CRO at a remote location. Financial criteria may then be applied and must include consideration of project management, transport of samples and travel costs, political stability within the country, availability of educated and well- trained staff. Location of the CRO in a country of choice with respect to ease of access to the location is also important to ensure uninterrupted shipment of samples and travel for sponsor scientists and QA personnel.
Fundamental to selecting a CRO is the conducting of a Quality audit. Ensuring accuracy, reliability and integrity is non-negotiable, and before a CRO is selected, the sponsor must conduct a review of Quality Systems operated by the vendor, to ensure compliance with regulatory agency expectations. Ideally, the vendor should have been audited by agencies such as FDA, EMEA and any observations fully addressed. Having determined that the necessary compliance systems are in place, the ability of the CRO to conduct the study must be assessed. This will include a review of the study size, available storage capacity of stability chambers, consideration of analytical equipment, ability to handle compounds with specific safety issues and the organization’s resources to complete testing within the required timeframe to keep the LTSS off the critical path for regulatory submission.
The sponsor and CRO must agree upon clear documented expectations for the investigation and reporting of OOT values. There should be no misinterpretation of the investigation process defined by the sponsor arising from differences in language, or delays due to time differences.
Only when these conditions have been met can cost and other business criteria be used to select a CRO from those vendors who can meet the aforementioned requirements.
Intellectual Property Protection
At the outset, the sponsor must put a confidentiality agreement in place with the CRO to protect against any breach of intellectual property surrounding the drug or product which is under development. In some low-cost countries, the national patent laws may not be as rigorous as in the sponsor country and it is expedient to protect the commercial asset under development. The vendor must be able to demonstrate strong corporate policies and procedures for IP protection
Development of the Study Protocol
For a successful study execution, detailed development and review of the stability protocol with the contract laboratory is critical, in order that the CRO gains a thorough understanding of the study plan, labelling, sample storage conditions, testing matrix, location of contingency samples if required, photo stability requirements and any other atypical testing that is to be conducted in the study. The stability protocol should be linguistically clear, concise and free from colloquial ambiguities arising from cultural or country-specific operating style differences.
Preparation for Initiating a Study
Thorough preparation is critical to successfully initiate a stability study, whether in-house or to a domestic CRO, however, outsourcing to a CRO overseas requires much greater detail in planning and preparation with added communication challenges (e.g., teleconference/videoconference) arising from geographical time differences and multilingual understanding and interpretation of the study expectations.
Clear communication regarding the turnaround times for each step of the process and a project plan with the timeline is critical for managing the outsourcing operation from start to finish as indicated by the RACI chart in Figure 2.
Initial meetings between an established CRO and the sponsor should occur approximately four months prior to the study start (this may be longer if CRO has not been previously contracted).
Steps should be taken to minimize any risk to the study by ensuring the CRO has a business continuity procedure, e.g., uninterrupted power supplies, alternative storage chambers, review frequency and follow up response to environmental monitoring chamber excursions, etc. in place in the event of a system failure which may have a higher risk in a low-cost developing country.
Following successful method transfer (typically taking approximately 26 days, range 20-45 days, n=5), QA audits and successfully addressing all major observations, and the delivery of samples for testing, the CRO can initiate the study ensuring that a fully documented procedure is available for chain of custody of samples through sample receipt, pull, testing, data entry and disposal.
Assessing Success
Executing the LTSS is one of the later steps in the commercialization of a new medicine, and there is no room for failure due to inadequate resources, quality compliance or poor scientific investigative practice. A successful study will be where quality data is generated within agreed timelines and budgetary costs.
Key performance indicators should be developed for testing to maintain the quality of data generated and efficiency of the study execution. From the outset, the CRO must be able to demonstrate that it is capable of testing all samples within the prescribed timeframe, having allocated sufficient resources to complete the task. This will also include the review and approval of laboratory data prior to its provision to the sponsor. A hallmark of a successful study will also be the clear documentation and reporting to the sponsor of any investigation of unexpected results, experimental observations or technical difficulties.
To drive a culture of continuous improvement requires careful definition, collection and review of metrics associated with the outsourcing process, and this should be applied to LCC outsourcing which may include
a) Performance; to understand where productivity gains could be made, e.g., by the provision of automated equipment or by streamlining lab operation, e.g., use of simple pre-printed lab notebooks to capture laboratory experimental observations and data reporting, or by developing best practices of faster data review, scheduling of equipment and laboratory staff, etc. If a procedure is automated, the manufacturer must be able to provide maintenance to support the technique, even at the remote location and this may influence a decision to use an automated method in preference to manual intensive procedures. The Bristol-Myers Squibb expectation is that testing will be completed within 30 days of sample pull. Our experience to date is that for timepoints where there are less than 10 samples, the typical delivery of approved data is about 12 working days, rising incrementally with the number of samples to be tested. For larger sample timepoints, the average delivery of data is 19 days (range 12-24 days). BMS expectations have always been met by CRO’s from LCCs.
b) Quality; evaluate the number and types of laboratory investigations into suspect or out-of-trend values. This may identify specific areas of vulnerability in the testing lab. This is, of course, contingent on trust being built so that all such values are reported upon observation. However, BMS’ experience of outsourcing to LCC has been positive with respect to quality. Recent BMS studies have provided a “right first time” quality index of 99.9%. Laboratory investigations identified the root cause of the majority of these suspect values to determinate failures such as column failure, contaminated glassware or poor precision of duplicate assays.
c) Cost; in order to become cost efficient, it is vital to compare the fully loaded costs of conducting studies at LCC with in-house or local (e.g., US or EU) studies. BMS have realized considerable savings through such outsourcing, with costs less than 50% of the in-house expenditure. When compared to local CRO’s the savings are considerably greater. Typically, fully loaded LCC costs are approximately 25% compared to US CRO’s. It is important to highlight that cost benefits are better if multiple studies are done at the same CRO with scientific staff that is common across studies.
The sponsor may wish to include quality control elements such as system suitability attributes monitoring to trend the performance of a specific method. On-going regular compliance data audits will also ensure the CRO is operating to the same high standards as at study initiation, and allows an opportunity to cultivate the partnership, discuss additional study requirements, optimized processes and shared benefits and improvements between the CRO and the sponsor. A higher level of compliance vigilance is advisable at a LCC CRO, compared to a domestic location, to ensure maintenance of quality standards through more frequent audits or assessments. The sponsor will also require an assurance that sample blinding and security procedures are in place to mitigate the risk of compromising intellectual property.
Post-Study Sample Management
At the conclusion of the timepoint or study, there should be clear instructions for the destruction of unused samples. The study protocol should be written so that there is minimal product remaining at the conclusion and returning this material to the sponsor should be unnecessary. Clear witnessed destruction must be fully documented by the CRO and copies provided to the sponsor of the study to prevent any unauthorized supplies being used illegally. Alternatively, unused samples should be returned to the sponsor when the final analytical data has been approved.
Conclusions
The outsourcing of stability studies has been routinely conducted by Pharmaceutical companies to create incremental internal capacity. Historically, the focus was to outsource to US- or EU-based CRO partners, however, with an increased focus on global development, global patient access and global clinical studies, BMS has decided to take advantage of well-established economical CROs in India and China to further reduce development cost and at the same time establish global presence in the growing economies.
This strategy is not entirely without risks and unexpected events may occur which require the sponsor to have a contingency plan, e.g., LCC may have a higher incidence of natural disaster, terrorism vulnerability, or local infrastructure deficiencies, which need to be considered. The strategic development plans of the CRO in such locations may also change, e.g., discontinuation of external CRO activities, and can lead to termination of the partnership agreement. This requires the sponsor company to either have alternate partners available, or to be able to react rapidly to identify and qualify a new alliance CRO.
Outsourcing repetitive data generation, which in itself has limited intellectual knowledge generation, has become part of a new Biopharma operating model focussed on bringing new medicine to patients as efficiently as possible, where vertically integrated models of the past are being replaced with concepts of selective integration. At BMS, selective integration with external collaborators is becoming an integral part of our business model, and hence, has developed strategic partners in scientifically capable but currently relatively low-cost countries to generate LTSS for new chemical entity regulatory submissions. Cost savings for the period 2007 to 2010 are estimated at $8 million, which is estimated to be 40% reduction in expenditure. This is based on eight large drug product LTSS conducted at a strategic partner and includes multiple strength products, some of which are single entity tablets, and others are fixed dose combination products (both immediate and extended release tablets). The data was provided to BMS without any compromise to regulatory filing dates, and the data itself has been accepted by several international regulatory agencies, including FDA.
The current BMS model is now to outsource all LTSS to off-shore CRO’s in India and China unless there is a strategic driver to keep the study in-house. This has enabled the redeployment of internal resources to other development roles within the organization to discover, develop and deliver innovative medicines that help patients prevail over serious diseases.
Tila Balasuriya is Group Leader responsible for long-term stability testing at BMS Moreton, UK. Recent experience includes outsourcing of stability studies overseas to contract facilities, delivery of analytical data packages for global registration stability studies, method transfers and validation of automated methodology.
Graham Clarke is Director of Analytical R&D for BMS at Moreton, UK where he is responsible for analytical activities to support the development of API and drug product including the strategic testing of long-term stability studies in low-cost countries. He received his Ph.D. from the University of Liverpool in 1981, and is a Fellow of the Royal Society of Chemistry.
Andrea Panaggio is Director of the R&D Stability group at BMS, New Brunswick, NJ. which supports stability of drug substance and drug product for small molecules and biologics from Phase I development to global registration. She received her Ph.D. from the University of Rhode Island in 1983 and is a member of the American Pharmacist Association and the AAPS.
Barry Scheer, Ph.D., is an Associate Director of Analytical R&D in the Pharmaceutical Development division of BMS at New Brunswick, NJ where he has responsibility for providing analytical development activities for life-cycle development projects. He also has considerable experience in successfully outsourcing long-term stability studies to India and China.
Pankaj Shah, Ph.D. is an Executive Director of Analytical R&D in the Pharmaceutical Development division of BMS at New Brunswick, NJ. He is responsible for analytical development activities of new chemical entities from early development to life-cycle management in support of API, DP, stability and non-invasive PAT activities for DP. He is also responsible for strategic outsourcing of analytical support activities globally.
Peter Timmins is an Executive Director of Biopharmaceutics R&D in the Pharmaceutical Development division of BMS with responsibilities at Moreton, UK and New Brunswick, NJ. He is responsible for the formulation of new drug products with specific emphasis on compounds which present significant challenges.
This article was printed in the September/October 2010 issue of Pharmaceutical Outsourcing, Volume 11, Issue 5. Copyright rests with the publisher. For more information about Pharmaceutical Outsourcing and to read similar articles, visit www.pharmoutsourcing.com and subscribe for free.