In order to maintain product quality, safety and efficacy during distribution, Good Distribution Practices (GDPs) specify that temperature-sensitive products are to be distributed (stored, handled and transported) throughout the distribution network (manufacturer, service providers and customers) as per specified temperature conditions. While GDPs have been in place for some time, recent regulatory scrutiny, industry initiatives and products requiring specific temperature conditions demonstrate the need to establish a company strategy for the distribution of temperature-sensitive products in accordance with product requirements, regulations and industry standards.
The initial focus of cold chain management should be the development and implementation of the cold chain shipment conformance process and the cold chain management processes (quality system) necessary to meet product and regulatory requirements. Unlike product storage in qualified temperature-controlled facilities, product shipping, even in qualified temperature-controlled shipping systems, is subject to a greater number of temperature excursions due to transportation process variability (ambient temperature, route, mode, transit time and in-transit handling). Consequently, temperature excursions occur during product shipping and although these temperature excursions are generally supported by product stability data, some countries enforce strict conformance with product label claim temperature. The implication of this enforcement, at the very least, is the risk of supply chain interruption due to shipment release delays and/or shipment rejections due to temperature non-conformances. Consequently, the initial focus of cold chain management should be the development and implementation of the cold chain shipment conformance process and the cold chain management processes (quality system).
While references may be to “cold chain,” the approach outlined applies to any temperature-sensitive product category (frozen, refrigerated, controlled-room temperature).

Figure 1 - Shipment Conformance Improvement Process
Cold Chain Shipment Conformance
A process for periodic shipment conformance measurement and reporting should be established after appropriate qualification of temperature-controlled shipping systems and implementation of post-qualification temperature monitoring. Shipment conformance is the basis for evaluating not only the continued performance of qualified shipping systems but also of the entire shipping lane process (origin packaging operation, transportation and destination receiving process). In order to measure shipment conformance and identify shipment conformance improvement opportunities, the following four-step process may be used (see Figure 1).

Figure 2 - Shipping Lane Compliance Model
Furthermore, the following shipping lane compliance model (see Figure 2) may be used as guidance for cold chain shipment conformance root-cause analysis and corrective action / preventive action (RCA and CAPA). This shipping lane compliance model is based on four root-cause categories (Shipping System, Pre-Transportation, Transportation, Post-Transportation), which provide for consistency in RCA and CAPA identification and implementation.
The cold chain shipment conformance process should be part of the overall cold chain management program, which in turn supports supply chain business objectives (compliance, customer service, cost). While improving shipment conformance to an acceptable level requires a structured problem-solving approach, it is the development and implementation of processes/procedures that ensure sustainability. Consequently, the next step in cold chain management should be the development and implementation of the cold chain management processes (quality system).

Figure 3 - Good Distribution Practices (GDPs)
Cold Chain Management Processes (Quality System)
The objective of the cold chain management processes (quality system) is to establish processes to maintain product quality and ensure compliance with global regulations and industry standards for the storage, handling and distribution of temperature-sensitive products. The cold chain management processes may be established with the starting point of Good Manufacturing Practices (GMPs) and Good Distribution Practices (GDPs). GMPs are not new, as they have been in effect for many years and are generally well understood. However, GDPs, while an extension of GMPs into the supply chain, are relatively new to manufacturers and especially to supply chain partners. A manufacturer’s control over the distribution network is much less than within manufacturing but manufacturers should establish quality and operational systems/processes (internally and with supply chain partners) to maintain product quality, safety and efficacy during distribution. These quality and operational systems/processes should include cold chain management, as well as import/export compliance and distribution control management (see Figure 3).

Figure 4 - Good Distribution Practices - Quality Systems

Figure 5 - Cold Chain Management - Operational Processes
With a GMP/GDP mindset and considering global good distribution practices and cold chain management guidance (such as WHO TRS 937 Annex 5 (Working Document QAS/08.252), 21CFR Part 210, 211 and 600, USP 1079, Canada Guide 0069, PDA Technical Report 39) and in accordance with the company’s quality systems, the cold chain management processes may be established. The objective of these cold chain management processes is to provide the foundation (quality system) to maintain product quality and ensure compliance with global regulations and industry standards for the storage, handling and distribution of temperature-sensitive products (see Figures 4-5).
While the initial focus of cold chain management should be on the development and implementation of the cold chain shipment conformance process and the cold chain management processes (quality system) necessary to meet product and regulatory expectations, there is potential to further strengthen cold chain management by developing a strategy to establish a shipping temperature range that is wider than the storage temperature range based on product stability. This is consistent with the industry trend of shipping outside label claim as supported by product stability and of filing this shipping temperature range. Regulatory agencies are also increasingly requesting stability data beyond long-term and accelerated stability data to support shipping temperature conditions outside label claim. There is already guidance from the EMEA on labeling statements supporting outside label claim temperature conditions, as indicated in CPMP/QWP/609/96/Rev 2 “Guideline on Declaration of Storage Conditions: A: In the Product Information of Medicinal Products B: For Active Substances” November 2007.
Distribution Product Stability Data Management
As already mentioned, unlike product storage in qualified temperature-controlled facilities, product shipping, even in qualified temperature-controlled shipping systems, is subject to a greater number of temperature excursions due to transportation process variability (ambient temperature, route, mode, transit time and in-transit handling). Depending upon the product, the shipping practice is to either ship per label claim (long-term storage temperature conditions using temperature-controlled shipping systems) or at ambient conditions (no temperature-controlled shipping systems are used). Consequently, a strategy for the distribution of temperature-sensitive products based on product stability should be developed. This strategy should include the development of guidance outlining the stability study conditions necessary to support a shipping temperature range that is wider than the storage temperature range. While there is global guidance (ICH Q1A(R2) 2003, WHO TRS 953 Annex 2 2009) on long-term and accelerated stability studies supporting typical storage temperature conditions, there is no industry or regulatory guidance on temperature excursion or temperature cycling stability studies to support outside label claim temperature conditions for a specified duration, such as during shipping. Consequently, this is an opportunity for the industry to define the temperature cycling stability study guidance to support a shipping temperature range that is wider than the storage temperature range.
While the focus of cold chain management should be on the development and implementation of the cold chain shipment conformance process, cold chain management processes (quality system) and product stability to support distribution, the ultimate objective is to facilitate good cold chain management practices throughout the distribution network.
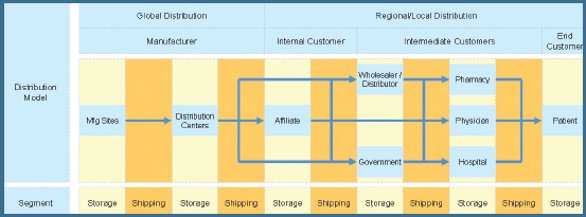
Figure 6 - Pharmaceutical Product Distribution Model
Cold Chain Management Throughout the Distribution Network
Good cold chain management practices throughout the distribution network may be instituted by partnering with service providers and customers (see Figure 6 for pharmaceutical product distribution model). Temperature-sensitive products are distributed (stored, handled and transported) within a distribution network comprised of manufacturer (manufacturing sites, distribution centers, commercial affiliates), service providers (carriers, freight forwarders, customs brokers, 3PLs/4PLs) and customers (wholesalers/distributors, governments, pharmacies, physicians, hospitals). In order to maintain product quality, safety and efficacy during distribution, it is important to raise awareness, communicate and educate supply chain partners as appropriate so that temperature-sensitive products are distributed in accordance with product requirements, regulations and industry standards. This may be done by assisting supply chain partners to establish processes to maintain product quality and ensure compliance with global regulations and industry standards within their distribution network.
Conclusion
In order to maintain product quality, safety and efficacy during distribution, Good Distribution Practices (GDPs) specify that temperature-sensitive products are to be distributed (stored, handled and transported) throughout the distribution network (manufacturer, service providers and customers) as per specified temperature conditions. Consequently, the initial focus of cold chain management should be on the development and implementation of the cold chain shipment conformance process and the cold chain management processes (quality system). The continued focus of cold chain management should be on product stability to support distribution, as well as partnering with service providers and customers to strengthen cold chain management throughout the distribution network.
Arminda Montero has worked in the pharmaceutical industry for over 15 years. She is the Distribution Quality Assurance Program Manager for the Global Pharmaceutical Products Division of Abbott Laboratories. Her current responsibilities include the development and implementation of strategies and quality systems for distribution, with specialized focus on cold chain management. Arminda holds a Bachelor of Science in Chemical Engineering from the University of Illinois at Urbana-Champaign.
This article was printed in the July/August 2010 issue of Pharmaceutical Outsourcing, Volume 11, Issue 4. Copyright rests with the publisher. For more information about Pharmaceutical Outsourcing and to read similar articles, visit www.pharmoutsourcing.com and subscribe for free.