Melissa Mooney- Director, eCOA Sales Engineering at IQVIA (eCOA Technologies)
In today’s highly connected healthcare ecosystem, wearable technology and electronic Clinical Outcome Assessments (eCOA) are converging to create new and unique opportunities for patient empowerment while at the same time driving the evolution of clinical research and treatment protocols. Patients are no longer bystanders - passive recipients of care – but engaged and active participants in their own therapeutic journeys.
However, legacy approaches that require patients to manage multiple devices and perform manual entry are cumbersome, placing heavy demands on patients and creating compliance problems that threaten study integrity. And with sponsors facing mounting pressure to reduce patient burden while obtaining superior-quality, real-time data, a workable solution was demanded that enables the clinical trials industry to simultaneously optimize patient burden, data quality, and operational efficiency to tackle highly prevalent medical conditions such as obesity and diabetes.
The integration of blood glucose monitors (BGM) with eCOA solutions address incumbent pain points in diabetes, obesity, and cardiovascular research to deliver quantifiable benefits across all stakeholder groups while enabling higher-quality scientific evidence. The burden on patients is significantly reduced as automatic triggering means that patients no longer need to remember to log hypo/hyperglycemic events manually. The net “win-win” is a dramatic improvement in compliance rates when data flows automatically from device to eCOA platform.
So Why Obesity?
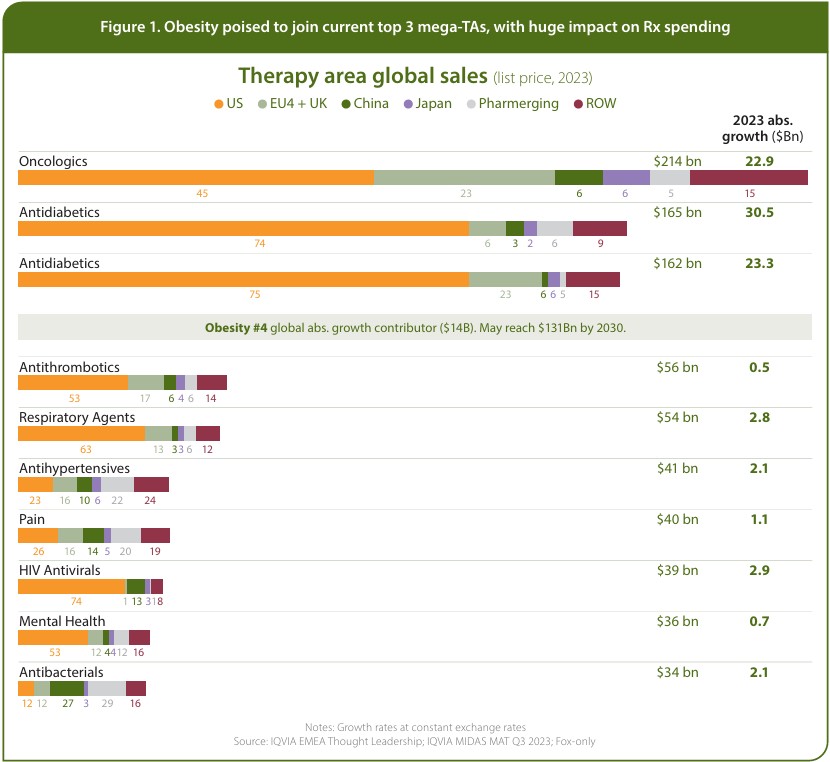
Obesity is a serious and increasingly prevalent condition that underpins a wide range of associated health conditions. As the obesity treatment market expands toward an expected $131 billion in 2030 (see Figure 1) - nearly ten times 2023’s $14 billion - the need for more sophisticated clinical trial designs is more important than ever. This phenomenal expansion in treatment is not only a testament to the breakthroughs in efficacy of GLP-1 receptor agonists like semaglutide and tirzepatide in trials but also stems from the global scale of the obesity epidemic.
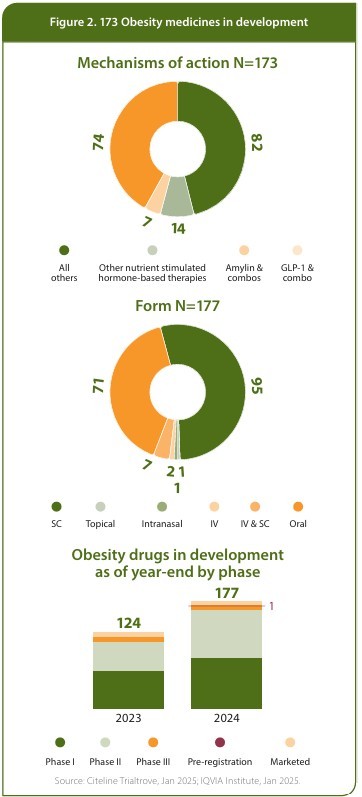
With 173 obesity medicines (see Figure 2) in the pipeline and regulatory agencies having to deal with numerous treatment choices, the bar for approval and reimbursement decisions keeps rising exponentially. This therapy area has expanded to become the world’s fourth-largest treatment category in terms of prescription spending, behind oncology, antidiabetics and immunology. However, with the enormous opportunity came a hyper-competitive development landscape presenting the pharmaceutical sector with as many giant-sized challenges as opportunities.
The Multi-Device Challenge
Modern clinical trials, particularly GLP-1 studies for diabetes and obesity, require patients to simultaneously manage multiple networked devices - glucose meters, blood pressure cuffs, activity monitors, ECG machines and wearables - while completing electronic questionnaires and maintaining long-term compliance.
This device proliferation creates substantial patient burden beyond mere inconvenience. Each requirement increases total burden and non-adherence risk, which is particularly problematic in obesity trials spanning months or years. Patient motivation fluctuates over time, especially during weight plateaus or when drug effects are slow and heterogeneous, leading to demoralizing setbacks despite drug efficacy.
Psychological burden compounds practical challenges. Obese patients typically enter trials after repeated weight-loss failures and with complex motivational dynamics that standard recruitment strategies cannot address. Unlike conditions susceptible to prompt symptom relief, obesity treatment demands prolonged vigilance around eating and weight management - potentially a problem for those with eating disorder histories or weight-related shame.
Traditional approaches that treat device readings and questionnaires as separate, post-hoc correlated data streams consistently fail. Patients must manually initiate questionnaires after glycemic events, enter device values, correlate BGM readings with diary entries and transfer meter data, a series of tasks that can create multiple error points, missing data and patient frustration, and compromise study integrity.
The Complexity of Modern Obesity Research
The broad comorbidity profile of obesity provides yet an additional layer of complexity that traditional research paradigms struggle to control to a satisfactory degree. Evidence suggests that with rising BMI, the incidence of comorbid conditions are significantly higher (see Figure 3): More than half of obese individuals develop cardiovascular disease or dyslipidemia, while about a third develop degenerative joint disease or type 2 diabetes.
This comorbidities profile provides a massive differentiation strategy for pharmaceutical companies marketing anti-obesity treatments. Instead of competing based only on weight-loss efficacy, which will not be sufficient to differentiate in an ever-more-saturated market, companies can sell their products as multi-mechanism therapies for the various aspects of obesity-driven health decline. But this requires data from several therapeutic categories traditionally obtained by having patients wear an overwhelming number of monitoring devices and completing gigantic questionnaire batteries.
The Integration Solution
Solutions that combine eCOA and BGM address the data collection burden on patients through automated, seamless correlation and capture of data. Instead of engaging patients with complicated questionnaires and manual data entry, electronic systems automatically initiate the necessary assessments when BGM readings hit pre-set thresholds.
When a patient’s glucose meter reading indicates a hypo glycemic or hyperglycemic episode, the eCOA system automatically prompts them to answer corresponding symptom questionnaires in near real-time or as soon as they can safely do so, with automated follow-ups as needed. 7 The goal is to get that information as close as possible to the event. Automatic triggering eliminates reliance on patients to remember when and how to record events. Instead, automated assessment drives compliance rates much higher while ensuring event information is being obtained in a timely manner - one less thing for patients to remember.
Technical underpinning for such solutions involves sophisticated data flow optimization that goes far beyond simple device connectivity. BGM values are automatically pulled into eSource systems and then subjected to protocol-specific logic for when questionnaire triggering is required. The event questionnaire is automatically linked to the BGM value by the system, precluding the potential for manual BGM entry and transcription errors that could compromise data quality and endpoint integrity.
This automated process brings dramatic time savings to sites and study teams who no longer must invest hours manually reconciling diary questionnaires with device data. eCOA and BGM data are housed together in eSource, where the site and study team can easily view merged data streams, and provides real-time electronic source data, reports and visualizations for unprecedented study visibility.
Real-World Applications and Proven Benefits
Streamlined patient experience in practice: Consider a traditional diabetes trial for which patients must take their glucose readings with a meter, write down the reading by hand, remember to fill out symptom questionnaires about hypoglycemic events and accurately copy meter readings onto diary pages. A combined BGM + eCOA solution transforms such a burdensome series of tasks into one of ease, with glucose values automatically recorded, numbers correlating to episodes of the hypoglycemic state identified in real-time, related questionnaires automatically initiated and all data going straight into the study database with proper temporal correlation.
The improvements to the patient experience are even more apparent: instead of dealing with several manual processes that take constant attention and memory, patients just use their glucose monitoring device in its normal way, but with the system automatically taking care of data capture and questionnaire prompting. This decrease in cognitive burden is especially beneficial for older patients or patients who have many chronic diseases to treat at the same time.
Device-agnostic platform integration: Advanced integrated solu tions provide device-agnostic eSource platforms that automate data collection from various manufacturers in a way that upholds regulatory requirements. The approach enables strategic device selection that balances rich data capture against participant experience without forcing patients to work with heterogeneous devices with different interfaces, software requirements and data protocols.
The platform solution addresses one of the largest operational challenges in modern clinical research: handling growing study complexity that necessitates several interdependent devices to support digital endpoints across indications. Instead of having patients contend with numerous devices and the surrounding software that already makes the process cumbersome, integrated platforms provide single interfaces that reduce training requirements and minimize the oppor tunity for user error.
Addressing Regulatory and Quality Considerations
The regulatory framework for digital health technologies has evolved significantly under clinical research, with agencies like the FDA providing increasingly more defined guidance on what criteria are acceptable for digital endpoints. Fully integrated BGM + eCOA solutions address most of the key regulatory concerns around digital health technologies, including data integrity, traceability, and validation.
Automated data capture eliminates a great deal of transcription errors and the data integrity risk of manual data capture procedures. Direct electronic capture of device data, with automatically prompted appro priate questionnaires, creates detailed audit trails that satisfy regulatory requirements for data traceability and source documentation.
Multi-stream data integration also supports more robust endpoint validation approaches. Instead of relying on isolated measures that may be subject to variability or measurement error, combined approaches enable cross-validation of results across diverse types of data, creating a stronger overall evidence package for regulatory filings.
Cost-Effectiveness and Operational Efficiency
Along with patient and data quality benefits, integrated solutions offer robust operational efficiencies that drive cost savings and improved study timelines. Data management processes at study sites require much less time, and automated data flow ends the need for tedious manual data entry, verification and reconciliation.
The real-time availability of combined data facilitates proactive manage ment of the study instead of reactive problem-solving. Study teams can identify compliance issues, data quality issues or patient participation issues in real-time instead of waiting for routine data review, making it possible to intervene and prevent study failure or data integrity problems that would compromise regulatory submissions.
Reports document stunning improvements in study teams’ ability to manage complex studies with more devices and questionnaires. Instead of spending considerable time training staff on various device platforms and data management protocols, sites can focus on patient care and clinical practices directly related to study objectives.
Patient Engagement and Long-Term Retention Strategies
The psychological cost of obesity research extends far beyond proce dural cost to include the emotional challenge of being in a constant state of vigilance for eating and weight management. Solution sets that can be embedded increase patient engagement by minimizing the administrative burden that can add to the psychological burden, allowing patients to focus on their care experience instead of struggling with complex data acquisition requirements.
The built-in automation within integrated systems also provides more accurate data capture when patient motivation wanes. Instead of patients remembering and filling out multiple manual steps during periods of difficulty, automated systems continue to gather relevant data even if patient involvement with voluntary activities has diminished.
This consistency is especially useful for long-term obesity trials, which require a delicate balance to maintain patient participation over months or even years. Balanced solutions provide sponsors with the richness of data collection without imposing unsustainable burdens on patient time and attention.
Future Implications and Industry Transformation
Integrated BGM + eCOA solution success in diabetes and obesity trials is powering broader transformation across the clinical research industry. As regulatory agencies become more comfortable with electronic endpoints and real-world evidence, smooth integration of many streams of data will increasingly be recognized for its value as it demonstrates treatment efficacy and safety across diverse patient populations.
The convergence of connected devices and eCOA platforms is not merely a tech advance - it is a necessary step toward patient-centered study design balancing participant experience with scientific rigor. It is particularly important as the industry attempts to keep up with increased competition for patient engagement and with the broader acceptance that patient burden has direct implications for data quality and study success.
New technologies like machine learning and artificial intelligence are beginning to increase the abilities of integrated platforms even more with predictive analytics capable of anticipating patient engagement issues and automatically switching data collection methodology to ensure compliance. Such technology will provide even greater study efficiency and patient experience gains.

Conclusion
The integration of eCOA platforms and BGM addresses the clinical research challenge of balancing comprehensive data capture with patient burden, offering measurable value while improving evidence quality. For obesity treatment researchers, integrated solutions convert manual procedures into automated workflows. This reduces burden, improving compliance rates, data accuracy, and long-term retention in extended studies.
Automatic association of device data and patient-reported outcomes creates real-time insights and cross-validation opportunities. Elimi nating manual transcription errors and providing robust audit trails strengthen data integrity while meeting regulatory expectations for digital endpoints. Study teams can redirect focus from labor-intensive reconciliation to meaningful patient care, while real-time data enables proactive study management.
As obesity treatment markets expand rapidly, efficiently capturing high-quality, multi-dimensional data becomes a critical differentiator. Integrated BGM + eCOA solutions enable pharmaceutical companies to demonstrate comprehensive therapeutic benefits across obesity-related comorbidities beyond weight-loss efficacy. Success in diabetes and obesity research accelerates broader industry transformation.
As AI and machine learning evolve, platforms will predict and adapt to patient engagement patterns, optimizing the balance between scientific integrity and participant experience. Integrated solutions represent key innovation for patient-centered study design. When patient burden directly impacts data quality and study success, these solutions offer a feasible path forward for all stakeholders. The question is no longer whether these solutions will be utilized, but implementation speed.
Melissa Mooney has over 20 years of experience in the development of eCOA solutions for use in clinical trials. Melissa’s area of expertise is eCOA solution design where she has supported clients and eCOA vendors in developing robust and usable eCOA software solutions that meet eCOA protocol requirements. She also brings a plethora of experience in eCOA requirement gathering, leading eCOA User Acceptance testing, eCOA data management, and BD support.